Need information on atomic radius trends? What's the trend for atomic radius? In this guide, we’ll clearly explain atomic radius trends and how they work. We’ll also discuss exceptions to the trends and how you can use this information as part of a broader understanding of chemistry.
Before we dive into atomic radius trends, let’s review some basic terms. An atom is a basic unit of a chemical element, such as hydrogen, helium, potassium, etc. A radius is the distance between the center of an object and its outer edge.
An atomic radius is one-half the distance between the nuclei of two atoms. Atomic radii are measured in picometers (one picometer is equal to one trillionth of a meter). Hydrogen (H) has the smallest average atomic radius at about 25 pm, while caesium (Cs) has the largest average radius at about 260 pm.
What Are the Atomic Radius Trends? What Causes Them?
There are two main atomic radius trends. One atomic radius trend occurs as you move left to right across the periodic table (moving within a period), and the other trend occurs when you move from the top of the periodic table down (moving within a group). Below is a periodic table with arrows showing how atomic radii change to help you understand and visualize each atomic radius trend. At the end of this section is a chart with the estimated empirical atomic radius for each element.
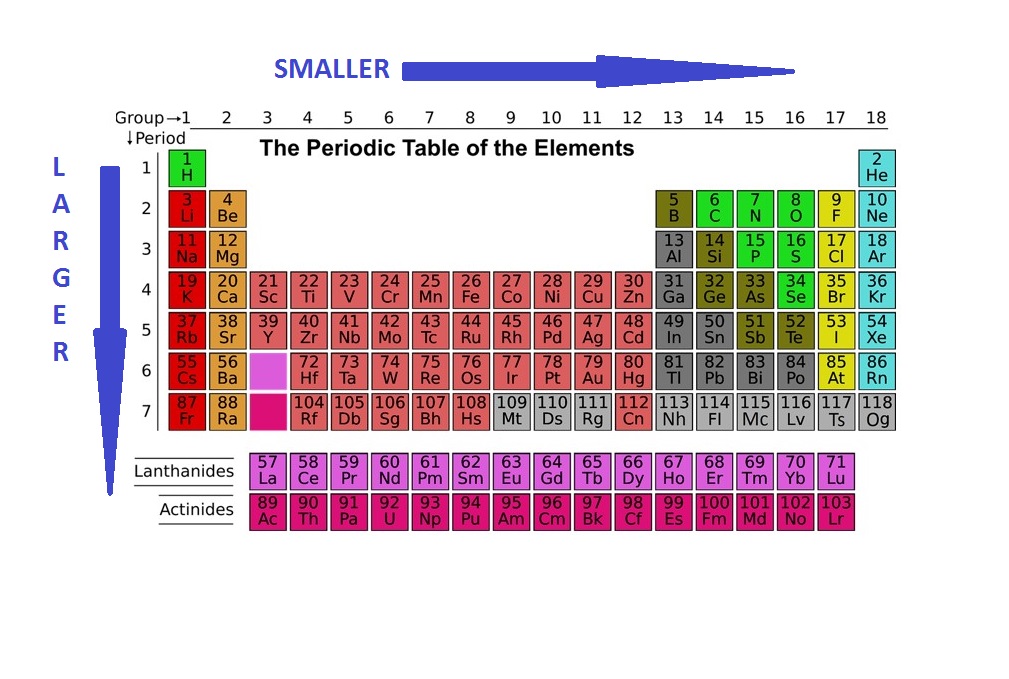
Atomic Radius Trend 1: Atomic Radii Decrease From Left to Right Across a Period
The first atomic radius periodic trend is that atomic size decreases as you move left to right across a period. Within a period of elements, each new electron is added to the same shell. When an electron is added, a new proton is also added to the nucleus, which gives the nucleus a stronger positive charge and a greater nuclear attraction.
This means that, as more protons are added, the nucleus gets a stronger positive charge which then attracts the electrons more strongly and pulls them closer to the atom’s nucleus. The electrons being pulled closer to the nucleus makes the atom’s radius smaller.
Comparing carbon (C) with an atomic number of 6 and fluorine (F) with an atomic number of 9, we can tell that, based on atomic radius trends, a carbon atom will have a larger radius than a fluorine atom since the three additional protons the fluorine has will pull its electrons closer to the nucleus and shrink the fluorine's radius. And this is true; carbon has an average atomic radius of about 70 pm while fluorine’s is about 50 pm.
Atomic Radius Trend 2: Atomic Radii Increase as You Move Down a Group
The second atomic radius periodic trend is that atomic radii increase as you move downwards in a group in the periodic table. For each group you move down, the atom gets an additional electron shell. Each new shell is further away from the nucleus of the atom, which increases the atomic radius.
While you may think the valence electrons (those in the outermost shell) would be attracted to the nucleus, electron shielding prevents that from happening. Electron shielding refers to a decreased attraction between outer electrons and the nucleus of an atom whenever the atom has more than one electron shell. So, because of electron shielding, the valence electrons don’t get particularly close to the center of the atom, and because they can’t get that close, the atom has a larger radius.
As an example, potassium (K) has a larger average atomic radius (220 pm)than sodium (Na) does (180 pm). The potassium atom has an extra electron shell compared to the sodium atom, which means its valence electrons are further from the nucleus, giving potassium a larger atomic radius.
Empirical Atomic Radii
| Atomic Number | Symbol | Element Name | Empirical Atomic Radius (pm) |
| 1 | H | Hydrogen | 25 |
| 2 | He | Helium | No data |
| 3 | Li | Lithium | 145 |
| 4 | Be | Beryllium | 105 |
| 5 | B | Boron | 85 |
| 6 | C | Carbon | 70 |
| 7 | N | Nitrogen | 65 |
| 8 | O | Oxygen | 60 |
| 9 | F | Fluorine | 50 |
| 10 | Ne | Neon | No data |
| 11 | Na | Sodium | 180 |
| 12 | Mg | Magnesium | 150 |
| 13 | Al | Aluminum | 125 |
| 14 | Si | Silicon | 110 |
| 15 | P | Phosphorus | 100 |
| 16 | S | Sulfur | 100 |
| 17 | Cl | Chlorine | 100 |
| 18 | Ar | Argon | No data |
| 19 | K | Potassium | 220 |
| 20 | Ca | Calcium | 180 |
| 21 | Sc | Scandium | 160 |
| 22 | Ti | Titanium | 140 |
| 23 | V | Vanadium | 135 |
| 24 | Cr | Chromium | 140 |
| 25 | Mn | Manganese | 140 |
| 26 | Fe | Iron | 140 |
| 27 | Co | Cobalt | 135 |
| 28 | Ni | Nickel | 135 |
| 29 | Cu | Copper | 135 |
| 30 | Zn | Zinc | 135 |
| 31 | Ga | Gallium | 130 |
| 32 | Ge | Germanium | 125 |
| 33 | As | Arsenic | 115 |
| 34 | Se | Selenium | 115 |
| 35 | Br | Bromine | 115 |
| 36 | Kr | Krypton | No data |
| 37 | Rb | Rubidium | 235 |
| 38 | Sr | Strontium | 200 |
| 39 | Y | Yttrium | 180 |
| 40 | Zr | Zirconium | 155 |
| 41 | Nb | Niobium | 145 |
| 42 | Mo | Molybdenum | 145 |
| 43 | Tc | Technetium | 135 |
| 44 | Ru | Ruthenium | 130 |
| 45 | Rh | Rhodium | 135 |
| 46 | Pd | Palladium | 140 |
| 47 | Ag | Silver | 160 |
| 48 | Cd | Cadmium | 155 |
| 49 | In | Indium | 155 |
| 50 | Sn | Tin | 145 |
| 51 | Sb | Antimony | 145 |
| 52 | Te | Tellurium | 140 |
| 53 | I | Iodine | 140 |
| 54 | Xe | Xenon | No data |
| 55 | Cs | Caesium | 260 |
| 56 | Ba | Barium | 215 |
| 57 | La | Lanthanum | 195 |
| 58 | Ce | Cerium | 185 |
| 59 | Pr | Praseodymium | 185 |
| 60 | Nd | Neodymium | 185 |
| 61 | Pm | Promethium | 185 |
| 62 | Sm | Samarium | 185 |
| 63 | Eu | Europium | 185 |
| 64 | Gd | Gadolinium | 180 |
| 65 | Tb | Terbium | 175 |
| 66 | Dy | Dysprosium | 175 |
| 67 | Ho | Holmium | 175 |
| 68 | Er | Erbium | 175 |
| 69 | Tm | Thulium | 175 |
| 70 | Yb | Ytterbium | 175 |
| 71 | Lu | Lutetium | 175 |
| 72 | Hf | Hafnium | 155 |
| 73 | Ta | Tantalum | 145 |
| 74 | W | Tungsten | 135 |
| 75 | Re | Rhenium | 135 |
| 76 | Os | Osmium | 130 |
| 77 | Ir | Iridium | 135 |
| 78 | Pt | Platinum | 135 |
| 79 | Au | Gold | 135 |
| 80 | Hg | Mercury | 150 |
| 81 | Tl | Thallium | 190 |
| 82 | Pb | Lead | 180 |
| 83 | Bi | Bismuth | 160 |
| 84 | Po | Polonium | 190 |
| 85 | At | Astatine | No data |
| 86 | Rn | Radon | No data |
| 87 | Fr | Francium | No data |
| 88 | Ra | Radium | 215 |
| 89 | Ac | Actinium | 195 |
| 90 | Th | Thorium | 180 |
| 91 | Pa | Protactinium | 180 |
| 92 | U | Uranium | 175 |
| 93 | Np | Neptunium | 175 |
| 94 | Pu | Plutonium | 175 |
| 95 | Am | Americium | 175 |
| 96 | Cm | Curium | No data |
| 97 | Bk | Berkelium | No data |
| 98 | Cf | Californium | No data |
| 99 | Es | Einsteinium | No data |
| 100 | Fm | Fermium | No data |
| 101 | Md | Mendelevium | No data |
| 102 | No | Nobelium | No data |
| 103 | Lr | Lawrencium | No data |
| 104 | Rf | Rutherfordium | No data |
| 105 | Db | Dubnium | No data |
| 106 | Sg | Seaborgium | No data |
| 107 | Bh | Bohrium | No data |
| 108 | Hs | Hassium | No data |
| 109 | Mt | Meitnerium | No data |
| 110 | Ds | Darmstadtium | No data |
| 111 | Rg | Roentgenium | No data |
| 112 | Cn | Copernicium | No data |
| 113 | Nh | Nihonium | No data |
| 114 | Fl | Flerovium | No data |
| 115 | Mc | Moscovium | No data |
| 116 | Lv | Livermorium | No data |
| 117 | Ts | Tennessine | No data |
| 118 | Og | Oganesson | No data |
Source: Webelements
3 Exceptions to the Atomic Radius Trends
The two atomic radius trends we discussed above are true for the majority of the periodic table of elements. However, there are a few exceptions to these trends.
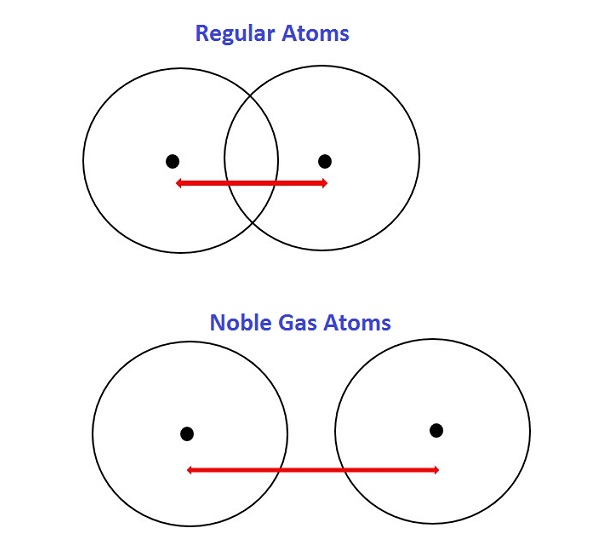
One exception is the noble gases. The six noble gases, in group 18 of the periodic table, are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). The noble gases are an exception because they bond differently than other atoms, and noble gas atoms don't get as close to each other when they bond. Because atomic radius is half the distance between the nuclei of two atoms, how close those atoms are to each other affects atomic radius.
Each of the noble gases has their outermost electron shell completely filled, which means multiple noble gas atoms are held together by Van der Waals forces rather than through bonds. Van der Waals forces aren't as strong as covalent bonds, so two atoms connected by Van der Waals forces don't get as close to each other as two atoms connected by a covalent bond. This means the radii of the noble gases would be overestimated if we attempted to find their empirical radii, so none of the noble gases have an empirical radius and thus don't follow the atomic radius trends.
Below is a very simplified diagram of four atoms, all about the same size. The top two atoms are connected by a covalent bond, which causes some overlap between the atoms. The bottom two atoms are noble gas atoms, and they are connected by Van der Waals forces that don't allow the atoms to get as close together. The red arrows represent the distance between the nuclei. Half of this distance is equal to atomic radius. As you can see, even though all four atoms are about the same size, the noble gas radius is much larger than the radius of the other atoms. Comparing the two radii would make the noble gas atoms look bigger, even though they're not. Including noble gas radii would give people an inaccurate idea of how big noble gas atoms are. Because noble gas atoms bond differently, their radii can't be compared to the radii of other atoms, so they don't follow atomic radius trends.
Other exceptions include the lanthanide series and actinide series at the bottom of the periodic table. These groups of elements differ from much of the rest of the periodic table and don’t follow many trends the other elements do. Neither series has a clear atomic radius trend.
How Can You Use This Information?
While you probably won’t need to know the atomic radius of various elements in your day-to-day life, this information can still be helpful if you’re studying chemistry or another related field. Once you understand each key atomic radius period trend, it makes it easier to understand other information about the elements.
For example, you can remember that noble gases are an exception to the atomic radius trends because they have a full outer electron shell. These outer electron shells also make the noble gases inert and stable. That stability can be handy. For example, balloons are typically filled with helium, not hydrogen, because helium is much more stable and therefore less flammable and safer to use.
You can also use atomic radii to estimate how reactive different elements will be. Atoms with smaller radii are more reactive than atoms with larger radii. The halogens (in group 17) have the smallest average radii in the periodic table. Fluorine has the smallest atomic radius of the halogens (which makes sense based on the trends), and that makes it highly reactive. Just adding fluorine to water will produce flames as the fluorine turns into a gas.
Summary: Periodic Trends Atomic Radius
There are two main atomic radius trends. The first atomic radius periodic trend is that atomic radii increase as you move downwards in a group. This is due to electron shielding. When an additional shell is added, those new electrons are farther from the atom’s nucleus, which increases atomic radius. The second atomic radius periodic trend is that atomic size decreases moving left to right across a period because the atom’s stronger positive charge due to having more protons attracts the electrons more strongly and pulls them closer to the nucleus, reducing the size of the atom.
There are a few exceptions to these trends, noticeably the noble gases which don’t form bonds the way most other atoms do, and the lanthanide and actinide series. You can use this information to better understand the periodic table, how atoms bond, and why certain elements are more reactive than others.
What's Next?
Need to brush up on your molecular chemistry? Review the different kinds of hydrates, how electronegativity works, and the uses (and limitations) of the Bohr Atomic Model.
Taking advanced chemistry and need some help? We have study guides for AP Chem and IB Chemistry, as well as a general Regents Chemistry review for New York high school students.
Dipping your toe into the wonderful world of biochemistry? Learn about the six types of enzymes and the chemical composition of nucleotides.